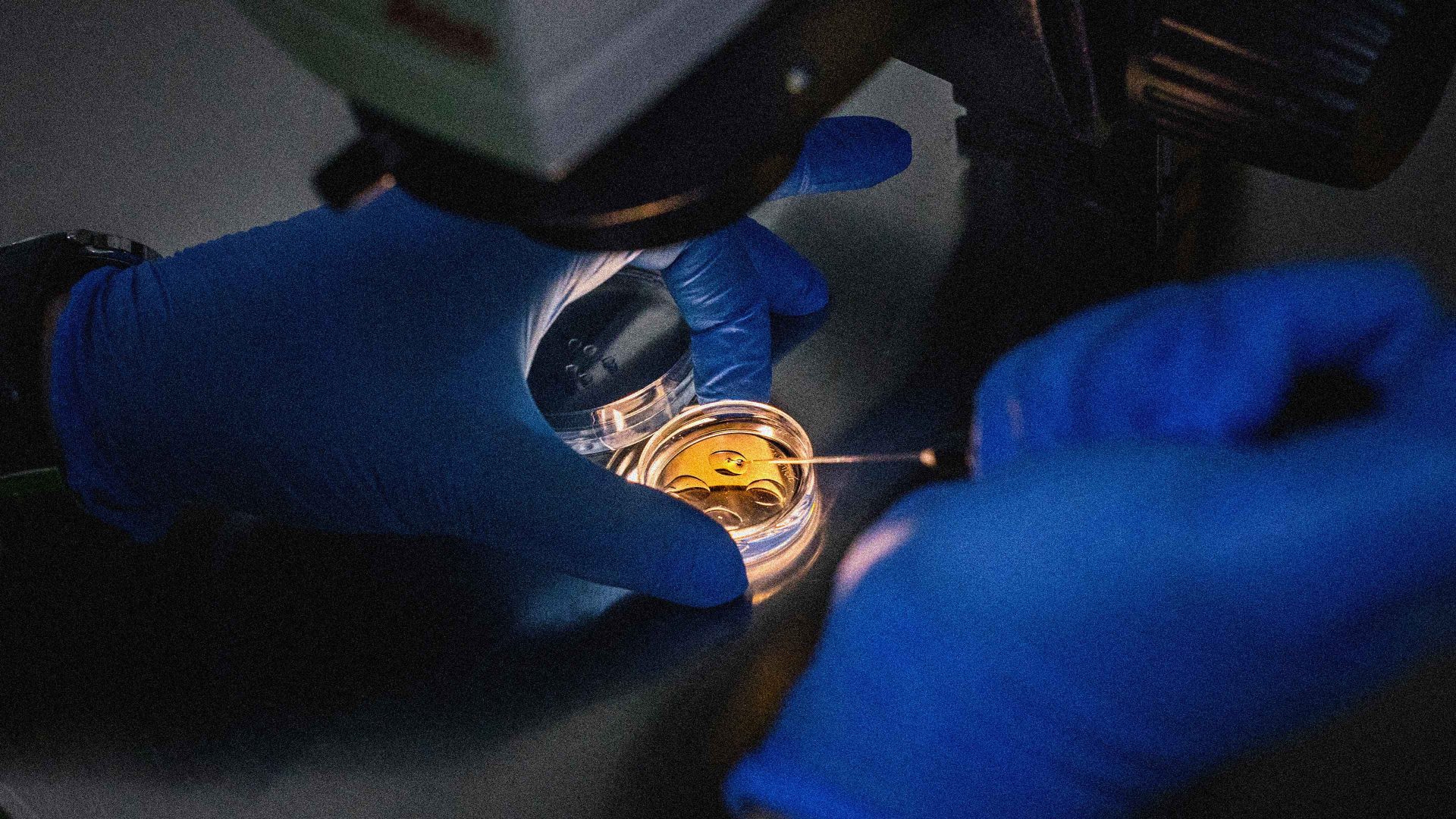
In vitro fertilisation (IVF) has been responsible for the births of more than 13 million children since it was first introduced almost 50 years ago. But it doesn’t solve all infertility problems. Some women produce very few eggs, and the quality of eggs declines with a woman’s age, lowering the chance of an IVF pregnancy. Some women lose their eggs in surgery for ovarian cancer, and men their sperm after prostate cancer treatment. Same-sex couples cannot produce children by IVF that are genetically related to both partners. And as it stands, egg collection is a painfully invasive procedure for a woman.
These problems could be mitigated by efforts to produce “artificial” gametes: sex cells (eggs and sperm) generated in the laboratory. But this remains a big challenge for human reproduction, not least because any such procedure would need to meet strict safety requirements – very low risk of producing developmental abnormalities in an embryo, say – before it could be approved for clinical use.
Experiments with mice have shown that it seems feasible in principle. The usual approach is to generate gametes in vitro from stem cells. It’s possible to guide embryonic stem cells – capable of producing all tissue types in the body – towards all manner of cell types, such as neurons, liver or muscle cells.
But gametes are harder to make because they are special: unlike other cells, they have only a single copy of our 23 chromosomes, so that only when the egg and sperm unite do the resulting cells have a full set of chromosome pairs. So gametes are produced not by regular cell division (mitosis) but by a special process called meiosis in which the chromosomes are carefully segregated.
Researchers have been able to conduct meiosis in mouse stem cells grown in a dish by first guiding the stem cells towards a cell type called primordial germ cells (PGCs) and then culturing these alongside ovarian or testicular cells which can guide the PGCs through the delicate process of meiosis, as they do in the gonads. The artificial gametes have then been used in IVF to produce mouse embryos that, when implanted in female mice, give rise to apparently healthy mouse pups.
Suggested Reading


2026: the year of the American basketcase
What’s more, we don’t need to start with embryonic stem cells. Pretty much any type of cell – a skin cell, say – can be transformed to a stem-cell-like state using a technique discovered in 2005. In this way, cells taken from a mature adult could in principle be used to make in vitro gametes.
But it hasn’t been so easy for human cells. Culturing human PGCs alongside ovarian cells can get them as far as a kind of egg-like precursor called oogonia cells, but not all the way to viable eggs.
Recently, a Japanese team led by “artificial gamete” pioneer Mitinori Saitou of Kyoto University showed that they could take mouse stem cells at least part of the way through the meiosis process – beyond oogonia – even without the help of ovarian cells. Not only does that simplify the process but it makes it easier to study the basic biology of egg production.
In September, a team at the Oregon Health and Science University (OHSU) reported a different approach to making “artificial” human eggs. The method is more akin to that used for cloning, first demonstrated in 1997 with Dolly the sheep. For cloning, you take a tissue cell – skin, say – and transfer its genetic material into an egg cell that has had its chromosomes removed, which can then grow into an embryo.
To make an egg cell rather than an embryo this way, you want to end up with only half the number of transferred chromosomes – 23 rather than 46 – in the egg, making sure you get one of each, so that the egg is capable of being fertilized by sperm. That’s the hard part, but the Oregon team managed it, after a fashion. They discovered a way to trigger cell division of eggs after receiving transplanted skin-cell chromosomes such that it mimics meiosis by throwing out half of the chromosomes.
Currently, this separation of chromosomes isn’t very reliable: many of the eggs end up with the wrong number of chromosomes, and the selection between them is random. The team made dozens of functional eggs this way which they then fertilised to make embryos. But fewer than 1 in 10 of the embryos could keep developing to the stage at which they would be transplanted for IVF, and even these were chromosomally abnormal and couldn’t even in principle have developed into healthy fetuses.
So at this stage it’s still not clear how viable the method would be as a fertility treatment, and all experts, including the researchers themselves, caution that it’s just a tentative start.